Core Concepts:
In this tutorial, you will learn how to read the periodic table. We will take a close look at the groups of the periodic table. In addition, you will learn about the different properties of the periodic table groups, periods, and families. If you enjoy this article, be sure to check out our others!
Related Articles:
- Periodic Trends Made Easy
- Elements
- How to Write Electron Shell Configurations
- Atomic Radius Trend
- Ionization Energy Trend
- Periodic Table with Element Names
Vocabulary
- Elements: A pure substance composed of a single atom with a unique atomic number.
- Groups: The vertical column of the periodic table that signifies the number of valence electrons in an element.
- Periods: The horizontal rows in the periodic table that signify the number of electron shells in an element.
- Families: Elements that have the same number of valence electrons and therefore similar properties.
View trends, families & groups interactively!
We think our periodic table is one of the best in the world! Visit our new interactive periodic table. You can view all sorts of trends, properties, magnetism, electrons, and even articles on all the elements!
You are watching: How to Read the Periodic Table
The Periodic Table and the Periodic Trends
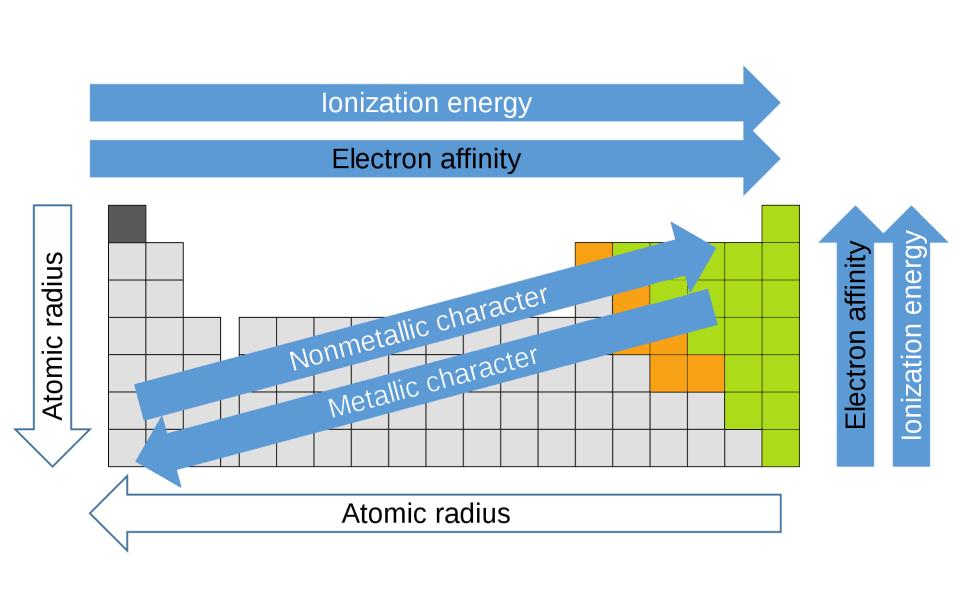
The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). Elements in the same group have the same number of valence electrons. Meanwhile, elements in the same period have the same number of occupied electron shells. In 1869 Russian chemist Dmitri Mendeleev noticed there existed an innate pattern of organization for the chemical elements. From this deduction, he formed the periodic table. It is important to note how the location of elements on this table tells us about their properties. A quick way to understand an element’s chemical and physical properties is to know the periodic trends. These trends tell you where the highest and lowest types of properties are concentrated on the periodic table. For a more in-depth explanation of periodic trends, click here.
Group vs Period
Groups are the columns of the periodic table, and periods are the rows. There are 18 groups, and there are 7 periods plus the lanthanides and actinides.
Periods on the Periodic Table
So what is a period on the periodic table? Periods are the horizontal rows of the periodic table. There are seven periods total and each element in a period has the same number of atomic orbitals. The top period, which contains hydrogen and helium, has only two orbitals. As you go down the rows, the number of orbitals increases. Below is a table to help visuals the periodic number and the corresponding orbitals.
Groups of the Periodic Table
See more : Electromagnetic Induction and its Applications
As previously mentioned, the vertical columns on the periodic table are called “groups”. There is eighteen groups on the periodic table in total, and each periodic table group contains elements with the same number of valence electrons.
The number of valence electrons present dictates the properties of an element. The reason for this is that the valence electrons, which are the electrons in the outermost shell, are the ones taking part in chemical reactions. These electrons are either donating, accepting, or sharing. Moreover, the more filled the valence shell is, the more stable the element.
How many groups are in the periodic table?
There are 18 groups in the periodic table, one per each column of the periodic table. The first column on the left is group 1, and the last column on the right is group 18.
Groups and Valence Electrons
The first group is the least stable as it only has one valence electron. Meanwhile, group eighteen is the most stable as these elements have a full valence shell (eight valence electrons). Below is a table relating the group numbers to the number of valence electrons.
Families of the Periodic Table
On the periodic table, there are families which are groups of elements with similar properties. These families are alkali metals, alkaline earth metals, transition metals, post-transition metals, metalloids, halogens, noble metals, and noble gases. Many of these families belong to a single group on the periodic table. However, not all of the families overlap with periodic table groups. For example, the transition metals contain all elements from group three to group twelve. Below is a periodic table where displaying the location of each family. You can also get a tutor to learn more about the table and see examples of several real elements.

See more : University Physics Volume 1
The name halogen means “salt formers” in greek. This is evident in nature as halogens interact with metals to form various salts. On another note, the halogens are a unique group of elements. They are the only periodic family that contains elements in the three states of matter at standard temperature. There are 6 halogens and they are located in group 17. These elements include fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). They are highly reactive, highly electronegative, and highly toxic non-metals.
Noble Metals
The noble metals consist of ruthenium (Ru), osmium (Os), rhodium (Rh), iridium (Ir), Pd, platinum (Pt), gold (Au), silver (Ag). Like the noble gases, they are inert due to having a complete valence shell. In addition, noble metals have catalytic tendencies. Also, they are very resistant to corrosion, tarnishing, and oxidation. Finally, like many of the other metals, they are soft and ductile.
Noble Gases (Group 18)
The noble gases, also called aerogens, are inert gases. Some examples include argon, krypton, and neon. They can be found in group eighteen on the periodic table. Likewise, this means they have a complete valence shell. For this reason, they are stable and relatively unreactive. Furthermore, the noble gases have low boiling points and low melting points. Physically they are colorless and have no smell.
See some cool Elements
Summary Table for Family Properties
See a Cool Chemistry Experiment
Further Reading
The Structure of an Atom
Periodic Trends Made Easy!
First 20 Elements of the Periodic Table
Source: https://en.congthucvatly.com
Category: Blog









